Lovo® Applications
Lovo works for any manufacturing process , achieve flexibility with a single instrument



Immunomagnetic Selection Prep
| Measure | Result | |
| Source Products | Collections | n=5 |
| Volume | 163.3 ± 28.5 mL | |
| Total Nucleated Cells (TNC) | 10.9 ± 3.4 x 109 | |
| Platelet Count | 4.46 ± 0.92 x 10 11 | |
| Processing Time | Automated Processing Time (With incubation) | 58 minutes |
| Max Processing Time | 61 minutes |
| Measure | Result | |
| 2 wash cycles to remove platelets and prepare for incubation | Platelet Depletion | 98.4 ± 1.0% |
| Targeted Pre-Incubation Volume | 100 mL | |
| Actual Pre-Incubation Volume | 100.6 ± 1.2 mL | |
| Full Procedure | TNC recovery | 97.2 ± 3.7% |
| TNC Viability | 96.3 ± 1.4% |
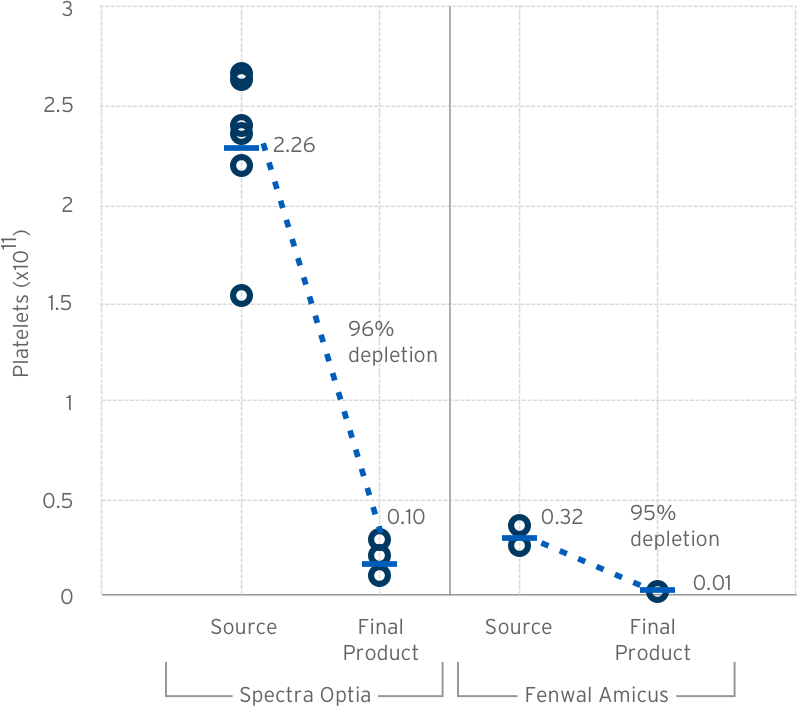
Fresh Leukapheresis Wash
| Measure | Result | |
| Collections (5L) | Fenwal Amicus | n=6 |
| Spectra Optia | n=8 | |
| Source | Volume | 90.8 ± 10.2 ml |
| TNC | 4.7 ± 1.8 x 109 | |
| Final Product | TNC Recovery | 98.6% |
| TNC Viability | 97.5% | |
| Target Final Volume | 95 mL | |
| Actual Final Volume | 95.3 ± 0.6 mL | |
| Processing Time | Average | 11:01 min |
| Maximum | 11:29 min |
Pre/Post Lovo Wash Platelet Count & Depletion
Culture Harvest & Media Exchange
Study objective
Compare T-Cell harvest methods
Goals
Reduce volume from 1.7L to 150mL
>99% (2 log) reduction of ancillary materials
High cell recovery
Maintain cellular CQAs
| Cell Type | Lovo
Fresenius Kabi | CS5+
Haemonetics | Manual |
| Recovery (%) | 93 ± 4 | 87 ± 3 | 91 ± 4 |
| Processing Time (min) | 30 ± 1 | 25 ± 4 | 61 ± 11 |
| Washout Efficiency (%) | 99.5 | 99.9 | 99.8 |
| Final Volume (mL) | 150 ± 1 | 153 ± 3 | 151 ± 6 |
| Cellular CQAs1 | comparable | ||
| Automated | ✓ | ± | x |
| Functionally Closed | ✓ | ± | x |
1 Critical Quality Attributes (CQAs) Measured: viability assessment, phenotype, cell functionality, ancillary materials.
Source: Lovo New Membrane Cultured Cell Testing Results: 223-DER-066723 – Data on file at Fresenius Kabi USA.
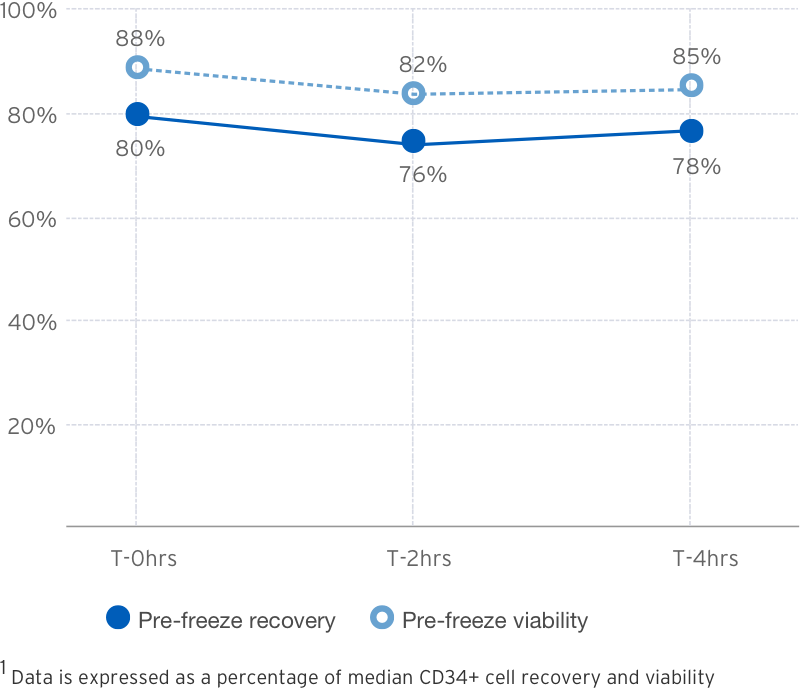
Thawed Wash & DMSO Removal
| Measure | Lovo 2.0
(three-cycle) |
| Number of Runs | 6 |
| PCV% | 8.4% (6.9 - 11.4) |
| Viable CD34+ Cell Recovery | 84% (61 - 93) |
| CD34+ Cell Viability | 92% (81 - 94) |
| DMSO Elimination | 97% (97 - 98) |
| Total Processing Time | 62 min |
† Data is expressed as median and (IQR)
Source: B.Mfarrej, et al. Pre-clinical assessment of the Lovo device for dimethyl sulfoxide removal and cell concentration in thawed hematopoietic progenitor cell grafts. Cytotherapy, Volume 19, Issue 12, 1501-1508.
Source: B.Mfarrej, et al. Pre-clinical assessment of the Lovo device for dimethyl sulfoxide removal and cell concentration in thawed hematopoietic progenitor cell grafts. Cytotherapy, Volume 19, Issue 12, 1501-1508.
Connect with an Expert for a Consultation
View Additional Lovo Resources
Lovo and Cue Field Service and Support Flyer
Lovo Integral to COVID-19 Lung Fibrosis Clinical Trial
Lovo Large Volume Cell Processing System Brochure
See how the Lovo® fits into the ScaleReady Workflow
The Lovo Cell Processing System is for laboratory use only and may not be used for direct transfusion. Appropriate regulatory clearance is required by the user for clinical use.
Refer to the Lovo Cell Processing System Operator’s Manual for a complete list of warnings and precautions associated with the use of these products.
Copyright © 2023 Fresenius Kabi AG. All rights reserved
REFERENCES:
- LOVO 2.X Blood Run Protocol Report: 223-DER-048957 – Data on file at Fresenius Kabi USA.
- LOVO 2.X Product Quality Test Results Design Review: 223-DER-048958 – Data on file at Fresenius Kabi USA.
- Lovo New Membrane Cultured Cell Testing Results: 223-DER-066723 – Data on file at Fresenius Kabi USA.
- B.Mfarrej, et al. Pre-clinical assessment of the Lovo device for dimethyl sulfoxide removal and cell concentration in thawed hematopoietic progenitor cell grafts. Cytotherapy, Volume 19, Issue 12, 1501-1508.
