We source Bio-Techne animal-free RUO and GMP-grade IL-2, IL-7, IL-15, and IL-21 liquid and lyophilized cytokines because they have the consistency, flexibility, quality, and regulatory support to facilitate the seamless scale-up of immune cell therapy manufacturing.
Focusing on cytokines for T cell and NK cell expansion, our GMP-grade cytokines boast robust reproducible bioactivity and flexible packaging formats that will accommodate closed-system requirements as you progress through clinical trials.
With ScaleReady, you’ll never need to worry if your RUO or GMP cytokines are truly animal-free. With industry-leading e. coli-derived animal-free recombinant protein manufacturing, our proteins minimize process risk by eliminating animal contaminants and mammalian pathogens.
Animal-free certificates of origin are available, and traceable batch records are kept for all our recombinant human RUO and GMP cytokines.
Smart sourcing of raw materials is necessary when building a commercially viable cell therapy manufacturing process. With high bioactivity and low lot-to-lot variability, Bio-Techne’s animal-free RUO and GMP cytokines can speed up your process development, lower manufacturing risk, and work favorably toward lowering overall manufacturing costs.
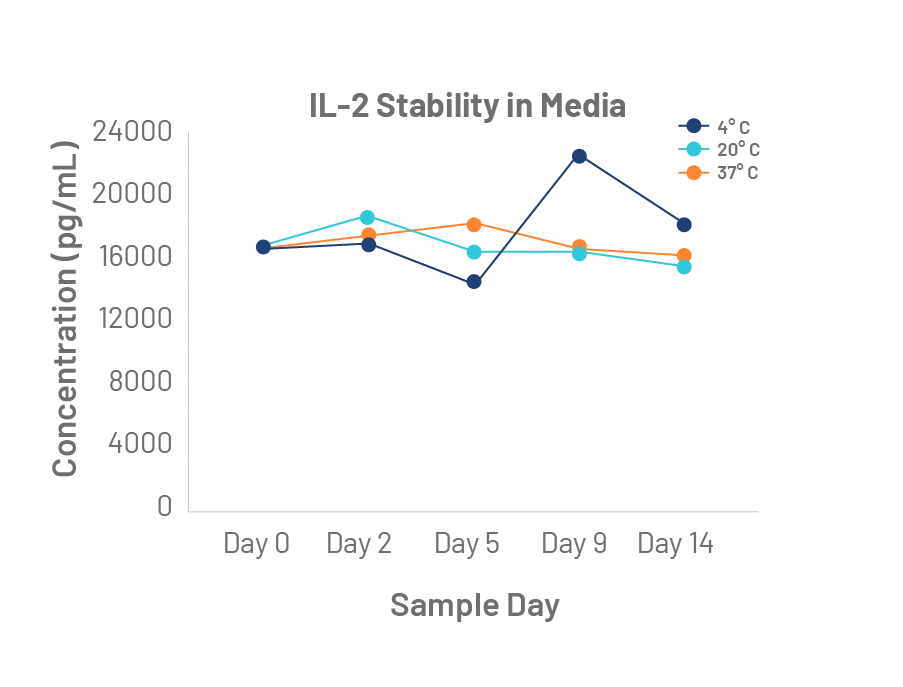
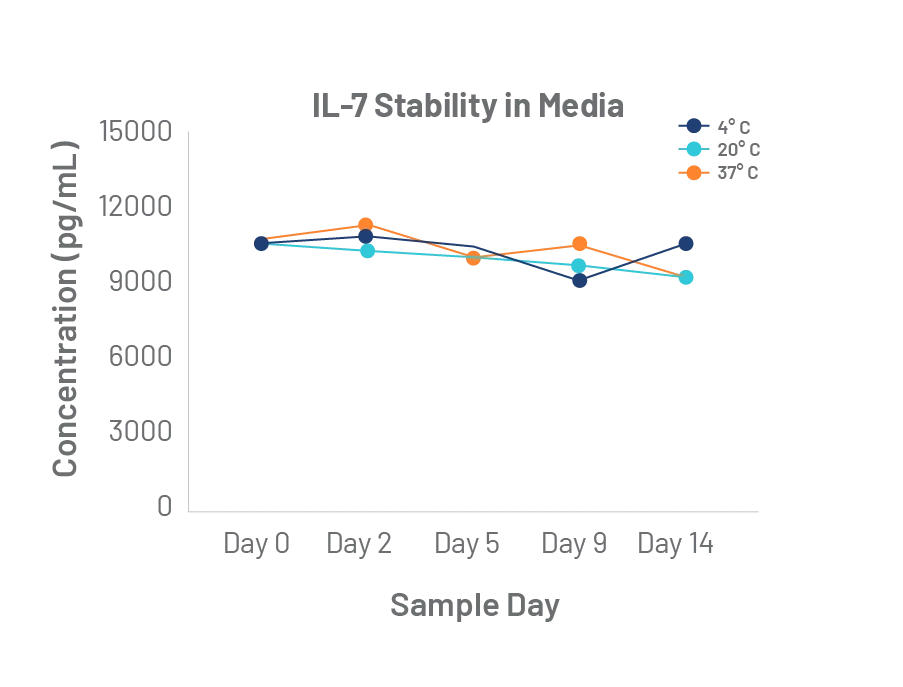
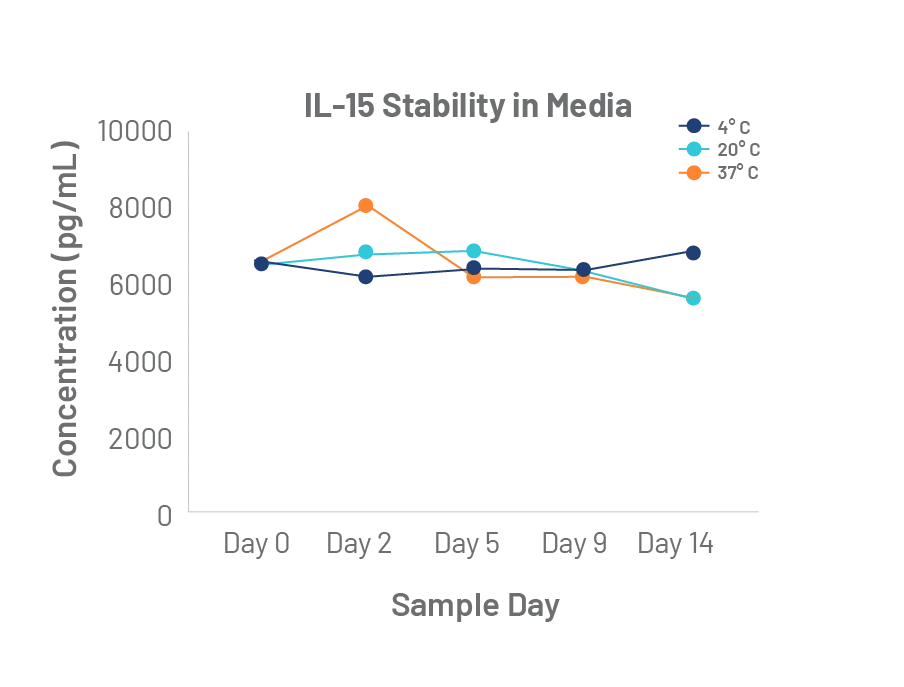
The quality of Bio-Techne cytokines is unmatched. Rigorous master cell lot control and lot-to-lot bioactivity testing, uniquely elevates the performance and consistency of our GMP cytokines to meet the long-term manufacturing requirements of the cell therapy industry.
Paired with Bio-Techne’s new large scale animal-free GMP manufacturing facility, the ScaleReady platform is prepared to provide a steady supply of cytokines throughout scale up and out of your cell therapy.
Now you can define the format of your raw materials. Bio-Techne GMP cytokines are available in off-the-shelf vials and custom mass/vial (liquid or lyophilized). All formats uphold rigorous quality and regulatory standards for GMP ancillary materials.
| GMP Protein | Source | DMF Filed | Format | Sizes | Catalog # | Research-grade catalog # |
|---|---|---|---|---|---|---|
| rhIL-2 | E. coli | Yes | Standard vial | 50 µg , 1 mg | BT-002-GMP | BT-002-AFL |
| 1 mg | ||||||
| Custom fill* | ||||||
| Standard Liquid Fill Vial | 50 µg , 1 mg | BT-002-GMP/LQ | BT-002-AFL/LQ | |||
| 1 mg | ||||||
| rhIL-7 | E. coli | Yes | Standard vial | 25 µg 25 µg, 1 mg | BT-007-GMP | BT-007-AFL |
| 1 mg | ||||||
| Custom fill* | ||||||
| Standard Liquid Fill Vial | 25ug | BT-007-GMP/LQ | BT-007-AFL/LQ | |||
| rhIL-15 | E. coli | Yes | Standard vial | 25 µg 25 µg, 1 mg | BT-015-GMP | BT-015-AFL |
| 1 mg | ||||||
| Custom fill* | ||||||
| Standard Liquid Fill Vial | 25ug | BT-015-GMP/LQ | BT-017-AFL/LQ | |||
| rhIL-21 | E. coli | Yes | Standard vial | 25 µg 25 µg, 1 mg | 8879-GMP | 8879-IL |
| 1 mg | ||||||
| Custom fill* | ||||||
