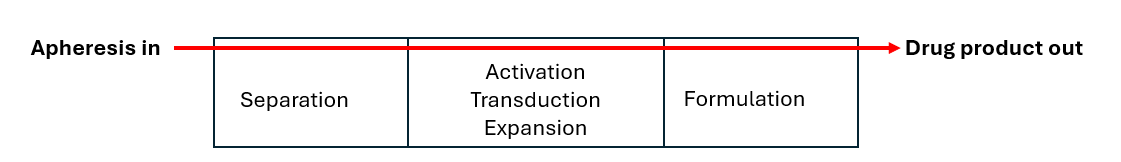
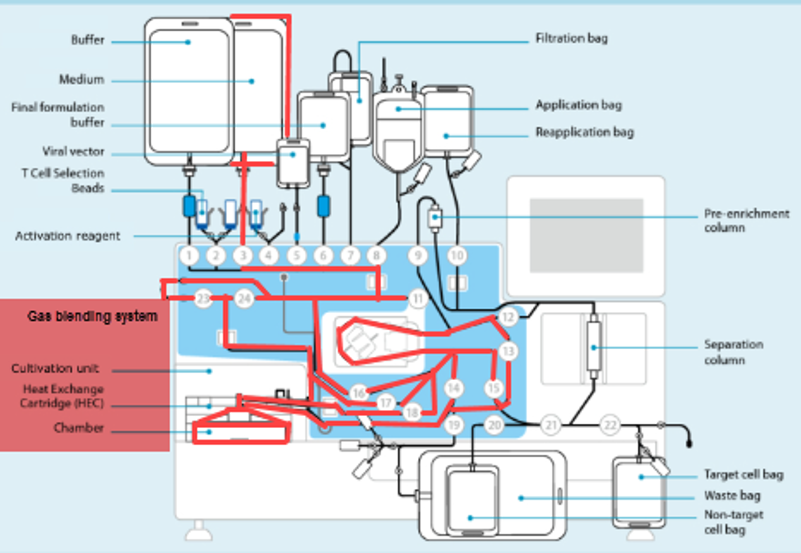
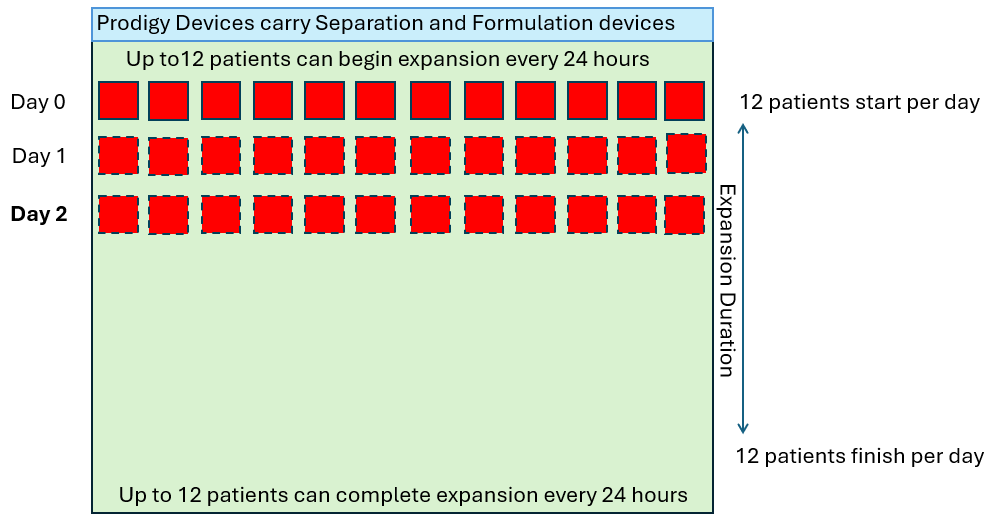
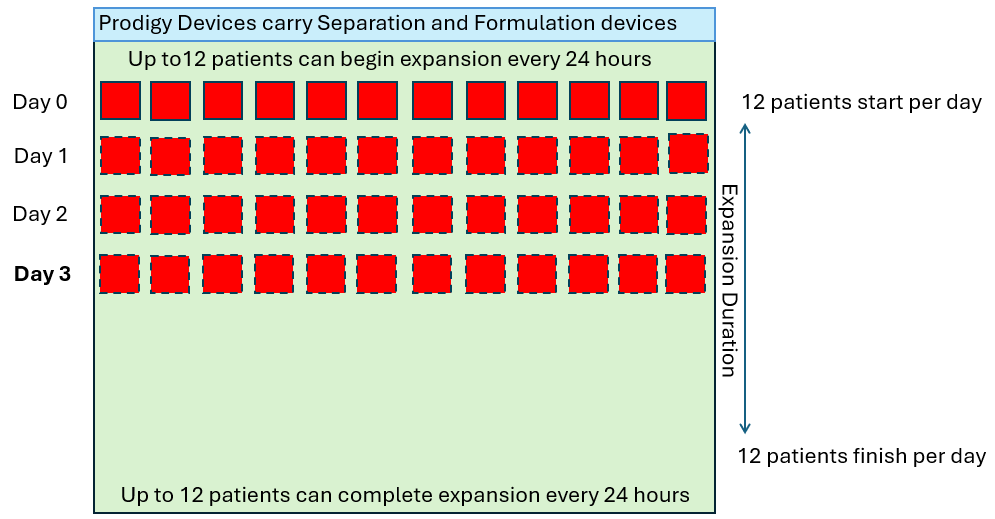
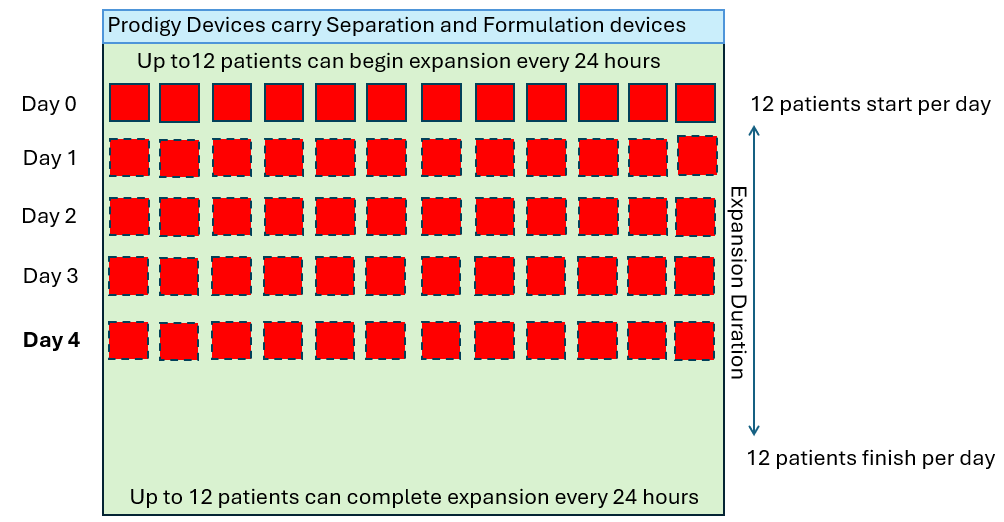
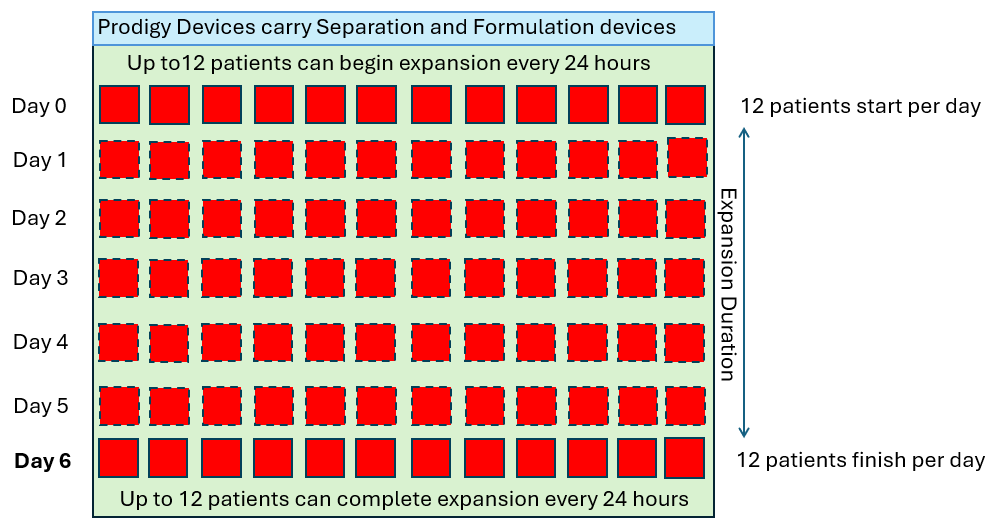
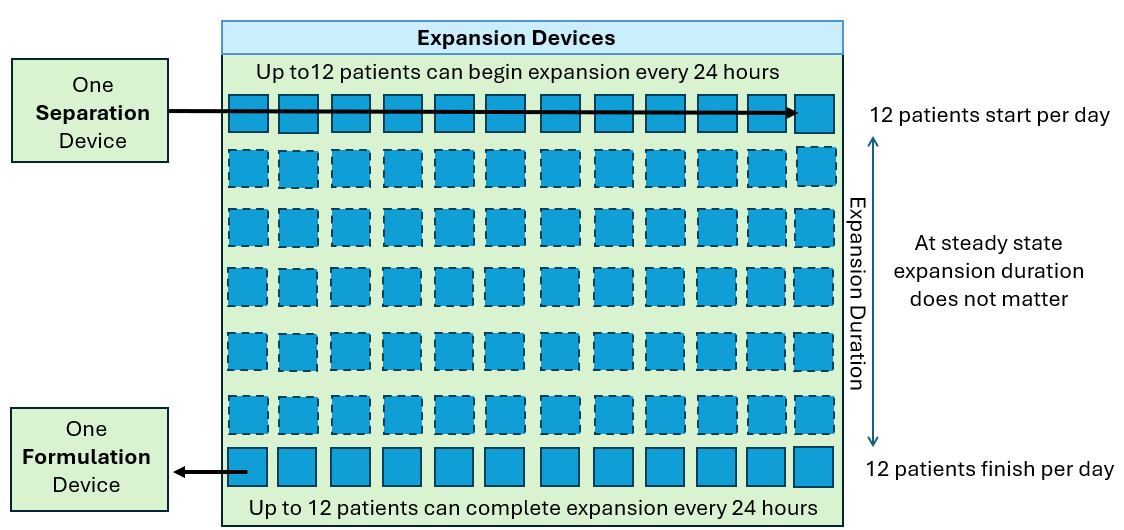
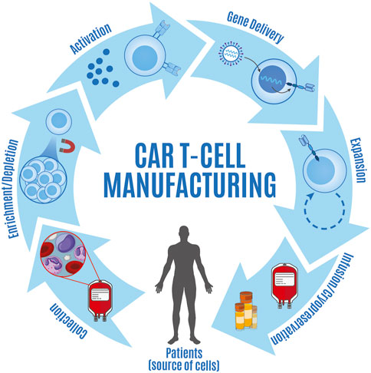
Single Use Metabolite Absorbing Resonant Tranducers (SMART)
In collaboration with Skroot Laboratories, SMART Sensors have been integrated into the G-Rex platform to provide developers and manufacturers of cell and gene-modified cell therapies (CGT) a non-invasive method of real-time, continuous monitoring of cell growth.
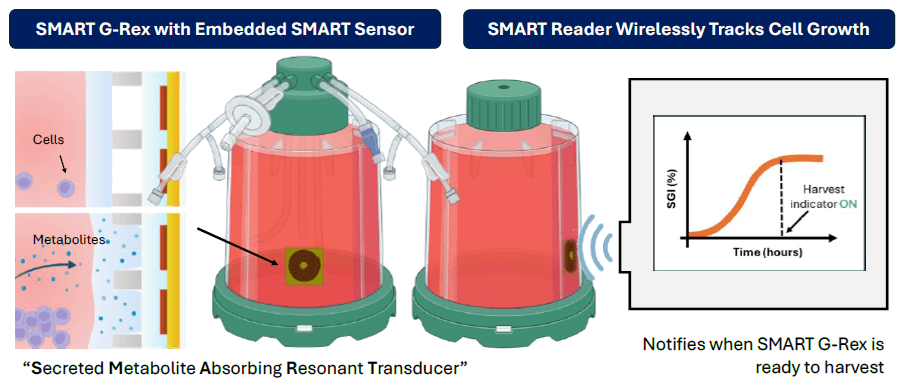
This integrated process analytic technology (PAT) can instantly report the extent of cell growth (or “report the phase of cell growth”) in a CGT drug production process and can inform manufacturers when a CGT drug product has reached the desired quantity of cells necessary to proceed with formulation, fill, and finish.
By integrating SMART technology, G-Rex is the only platform for CGT manufacturing that can avoid intervention when determining cell quantity or allow real-time monitoring of cell growth throughout the production run.
G-Rex100M-CS Sensor Enabled, Sterile Fluid Path
(P/N G281100-CS-SE)
*ETA Q4 2026*
How do SMART sensors work?
Resonant sensors are passive inductor-capacitor (LC) circuits whose measured resonant frequency shifts when the electrical permittivity of their immediate environment changes. Their utility for wireless monitoring in sealed or otherwise non-vented systems (where direct access to the interior is not practical or permitted) has been documented across several application areas [1,2]. In the specific context of cell culture monitoring, they enable noncontact interrogation of cultures without sampling. Recently, an advancement on the conventional resonant sensor called the SMART (single-use, metabolite absorbing resonant transducer) sensor was introduced [3]. The SMART design incorporates a polymer layer that softens in response to secreted metabolites, amplifying the local permittivity contrast above the conventional resonator and providing >20-fold higher sensitivity to growth dynamics than direct resonant sensing. Physically, the SMART sensor comprises five layers (from fluid-facing side to vessel-facing side): a transduction membrane responsive to secondary metabolites, a polyethylene terephthalate (PET) sheet with engineered voids, a bonding interlayer, a patterned LC resonator (copper coil on polyimide), and a mounting adhesive for vessel attachment (Fig. #a)[4].
As cells grow, secreted secondary metabolites (organic signaling molecules) selectively absorb into the SMART sensor membrane, softening the membrane material such that it partially collapses into engineered voids in the sensor structure (Fig #b). Prior screening work has shown that simple terpenoids, such as prenol, produce strong responses. For T cells, there is evidence of secreted lipid mediators, including eicosanoid derivatives (e.g., prostanoids and leukotrienes), as signaling molecules [5]. We have verified that arachidonic acid (AA), an omega-6 PUFA that is a precursor to eicosanoids, causes a robust sensor response (see SMART G-Rex publication [6]). This is representative of a broad class of secreted organic compounds that these cells produce during active growth that act as a plasticizer on the sensor membrane.
[1] Huang QA, Dong L, Wang LF. LC Passive Wireless Sensors Toward a Wireless Sensing Platform: Status, Prospects, and Challenges. Journal of Microelectromechanical Systems 2016;25:822–41. https://doi.org/10.1109/JMEMS.
[2] Carr AR, Chan YJ, Reuel NF. Contact-Free, Passive, Electromagnetic Resonant Sensors for Enclosed Biomedical Applications: A Perspective on Opportunities and Challenges. ACS Sens 2023;8:943–55. https://doi.org/10.1021/
[3] Chan YJ, Dileep D, Rothstein SM, Cochran EW, Reuel NF. Single-Use, Metabolite Absorbing, Resonant Transducer (SMART) Culture Vessels for Label-Free, Continuous Cell Culture Progression Monitoring. Advanced Science 2024;11:2401260. https://doi.org/10.1002/advs.
[4] Reuel NF, Chan YJ. Signal enhancement of resonant sensor for cell measurements. WO2024058838A1, 2024.
[5] Nicolaou A, Mauro C, Urquhart P, Marelli-Berg F. Polyunsaturated Fatty Acid-Derived Lipid Mediators and T Cell Function. Front Immunol 2014;5:75. https://doi.org/10.3389/fimmu.
[6] https://doi.org/10.1101/2025.
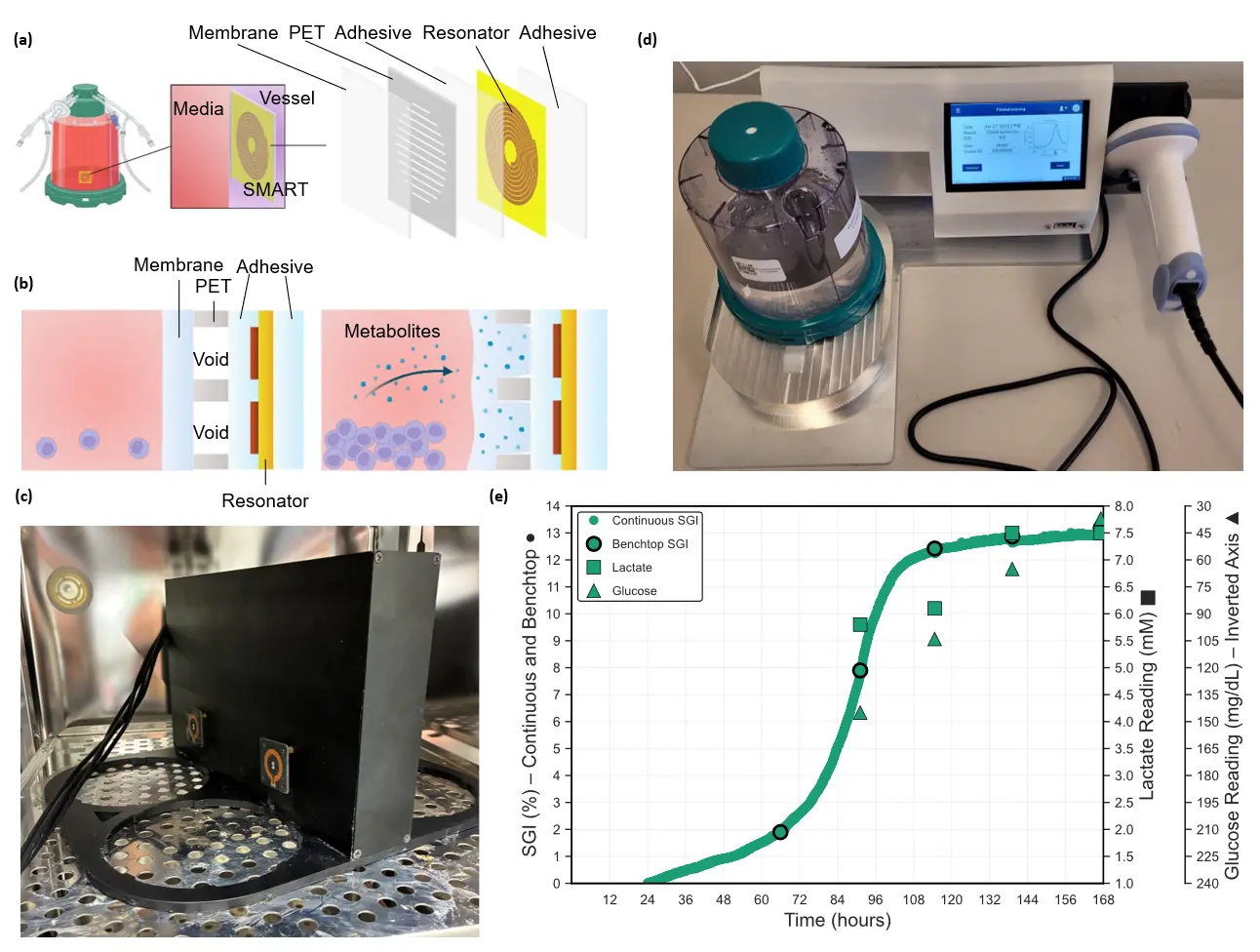
SMART sensor structure and response. (a) The SMART sensor sticker is adhered to the inside of the G-Rex vessel to be in contact with the growth medium and is comprised of five layers (from fluid side: responsive membrane, PET sheet with engineered voids, adhesive tie layer, resonator, and adhesive layer for bonding to vessel interior. (b) Sensor transduction mechanism: as the cells secrete secondary metabolites (terpenoids) the membrane softens, fills the voids and the local permittivity to the resonant coil changes, causing a change in resonant frequency. (c) Custom reader to interrogate the sensors continuously within an incubator. (d) Custom reader to interrogate the sensors intermittently in a bench-top reader mode. In both cases, the G-Rex is kept closed and the cell layer is undisturbed. (e) Response of sensor reported as the change in resonant frequency from the start point (Skroot Growth Index, SGI) in both continuous and benchtop mode compared to traditional
What does the FDA say about PAT?
Guidance for Industry
PAT — A Framework for Innovative Pharmaceutical Development, Manufacturing, and Quality Assurance
U.S. Department of Health and Human Services Food and Drug Administration
Center for Drug Evaluation and Research (CDER)
Center for Veterinary Medicine (CVM)
Office of Regulatory Affairs (ORA)
Pharmaceutical CGMPs
September 2004
In accordance with previously issued Guidance for Industry from the Food & Drug Administration (FDA), this new integrated process analytic technology (PAT) will help CGT developers and manufacturers:
- Design and develop well understood processes capable with higher degrees of predictability
- Consistently ensure the production of a CGT drug product of predefined quality (i.e. Quality by Design)
- Mitigate risk and variability associated with manual sampling
- Increase throughput and drug product supply by reducing production cycle times
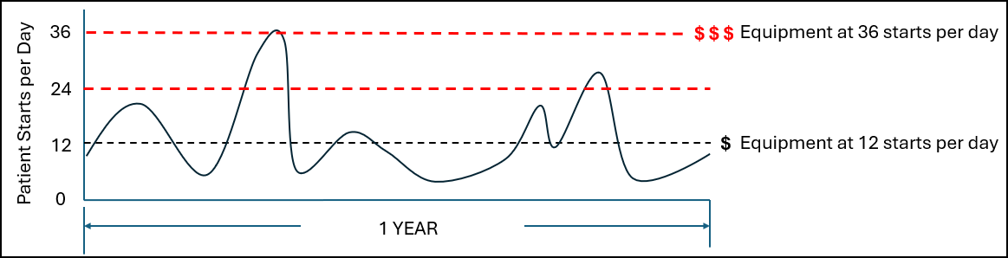
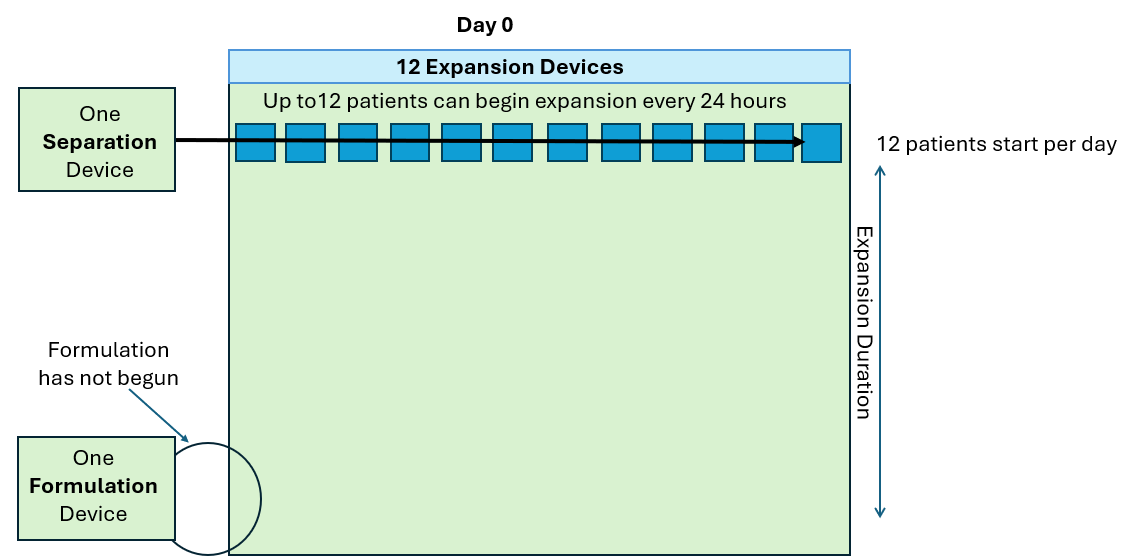
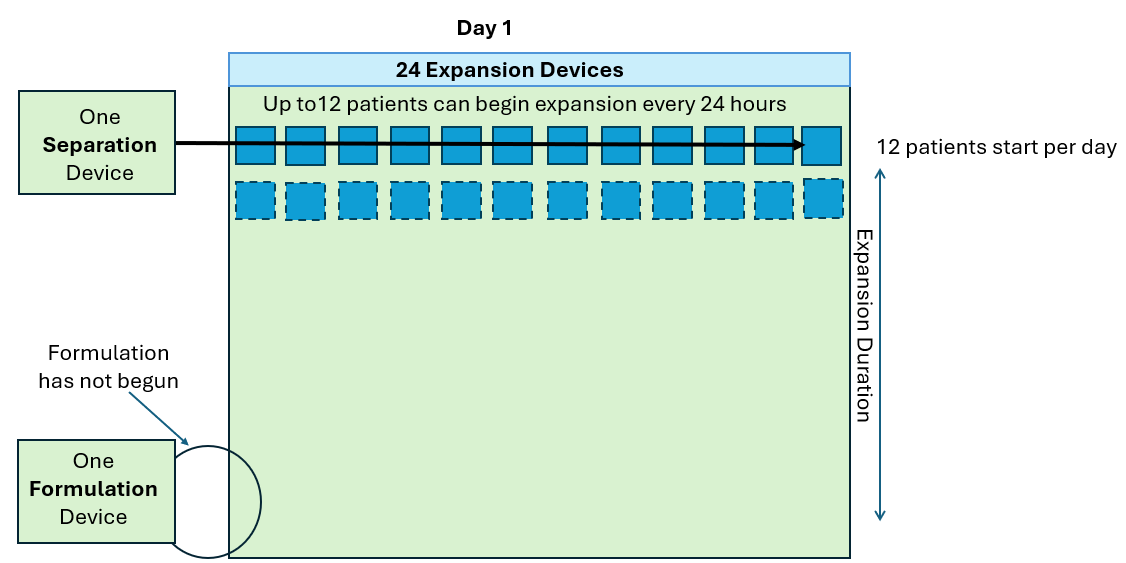
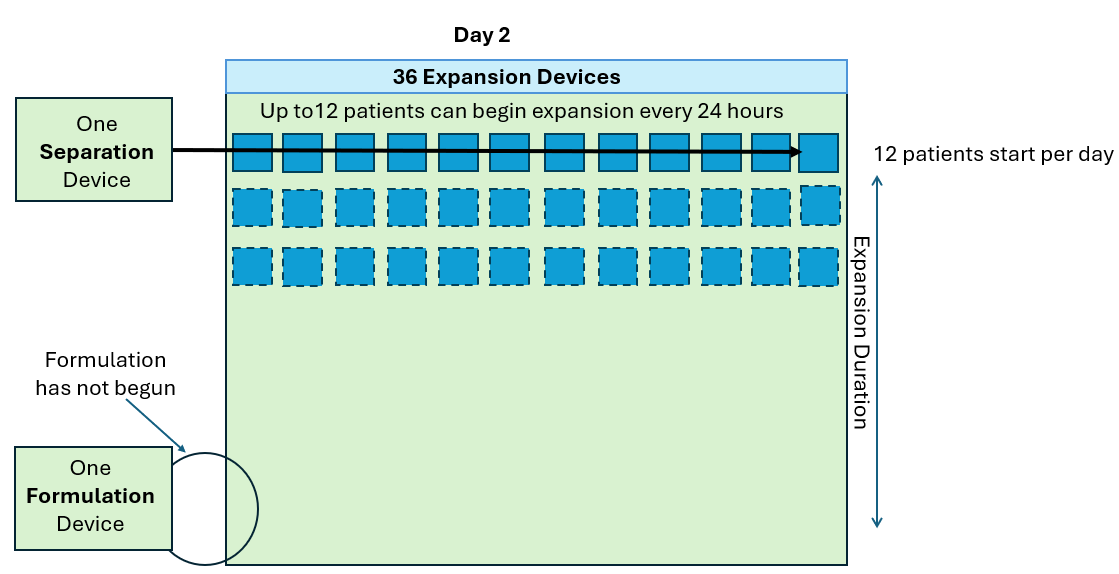
Work SMARTer, not harder!
Knowing the level of cell growth in your G-Rex can be useful in the following ways:
- Real-time confirmation when a patient’s dose requirements have been met triggering an action
- Early detection of “no growth” conditions triggering an action to inform the patient’s physician.
- Early detection of “aberrant growth” conditions triggering an action to inform the patient’s physician.
- Monitor cell growth extent without intervention (contact free, no sampling)
- Notify end of growth (optimal harvest point)
- Detect aberrant growth early (no growth or contamination events)
- Determine cell expansion level at end of growth (eliminate post cell count)
- Monitor remotely via online dashboard (with continuous reader)
The remote monitoring, AWS-powered dashboard is simple to use and can be customized or further integrated into standard ERP systems or eQMS software. Data from current and past production runs can be used to acquire critical insights previously unavailable.
Moreover, the ability to qualify a multi-drug product ballroom style of production is more of an operational problem than a regulatory one. The SMART G-Rex platform may also serve a crucial function in driving LEAN operations by providing manufacturer’s a simple way to “warehouse” many drug products within the same incubator while maintaining segregration, control, and traceability.

The incubator’s that are recommended for G-Rex production are Thermo Fisher’s Vios iDx 255L C02 incubators.

A bird’s eye view of SMART G-Rex devices configured in a 3×3 array of continuous readers.

A lateral view of SMART G-Rex devices configured in a 3×3 array of continuous readers.
A single incubator stack, like the ones seen above, will be able to comfortably accommodate 54 G-Rex100M-CS nestled next to a SMART continuous reader. This kind of density is unparalleled in cell & gene therapy manufacturing and will be a core driver of logarithmic reductions in drug product cost by amortizing less overhead over more drug products.
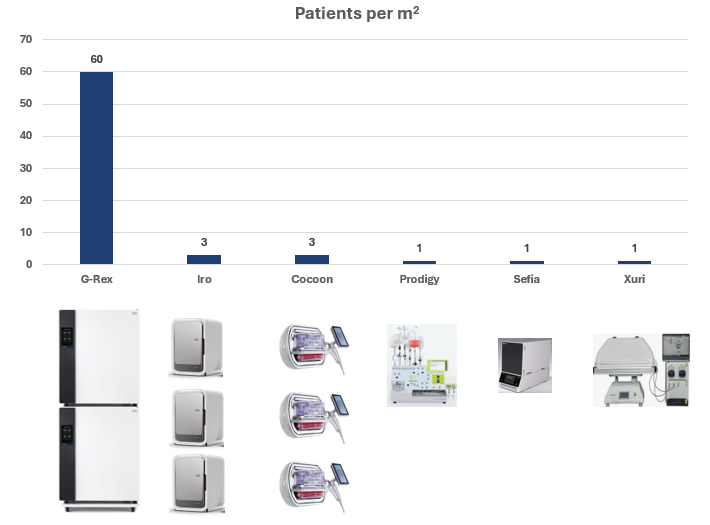
In summary, the simple nature of G-Rex has allowed it to become a sophisticated tool that is future-proofed because new innovations, such as SMART sensors, are easily incorporated into novel G-Rex designs without requiring us to fundamentally alter the G-Rex bioreactor design.