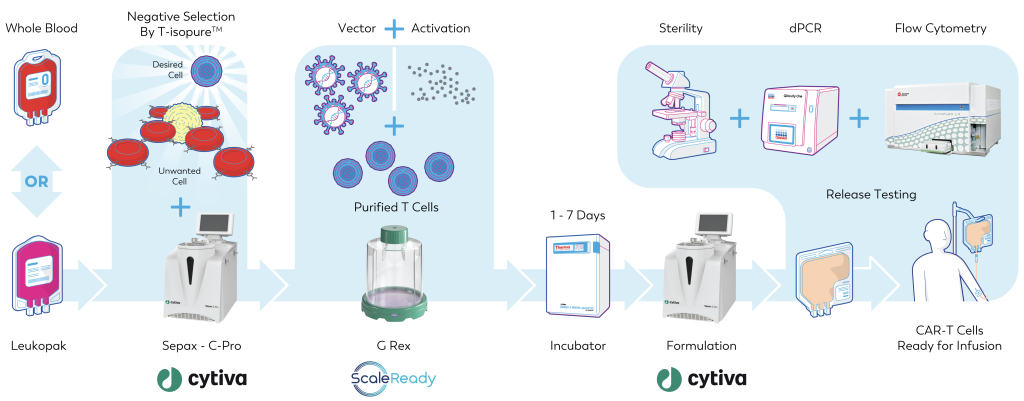
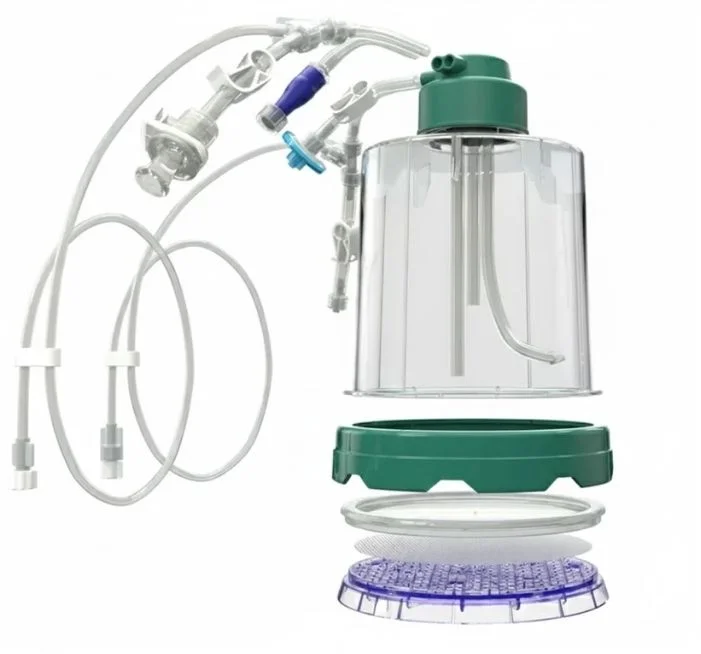
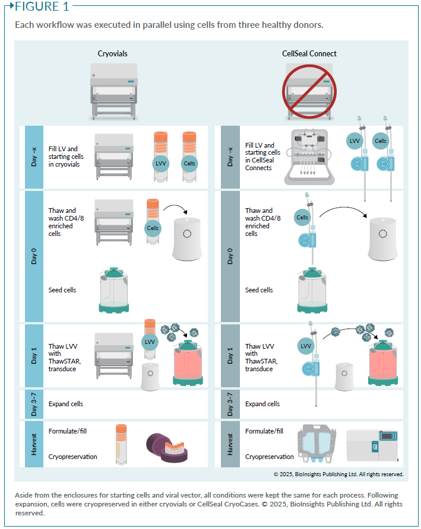
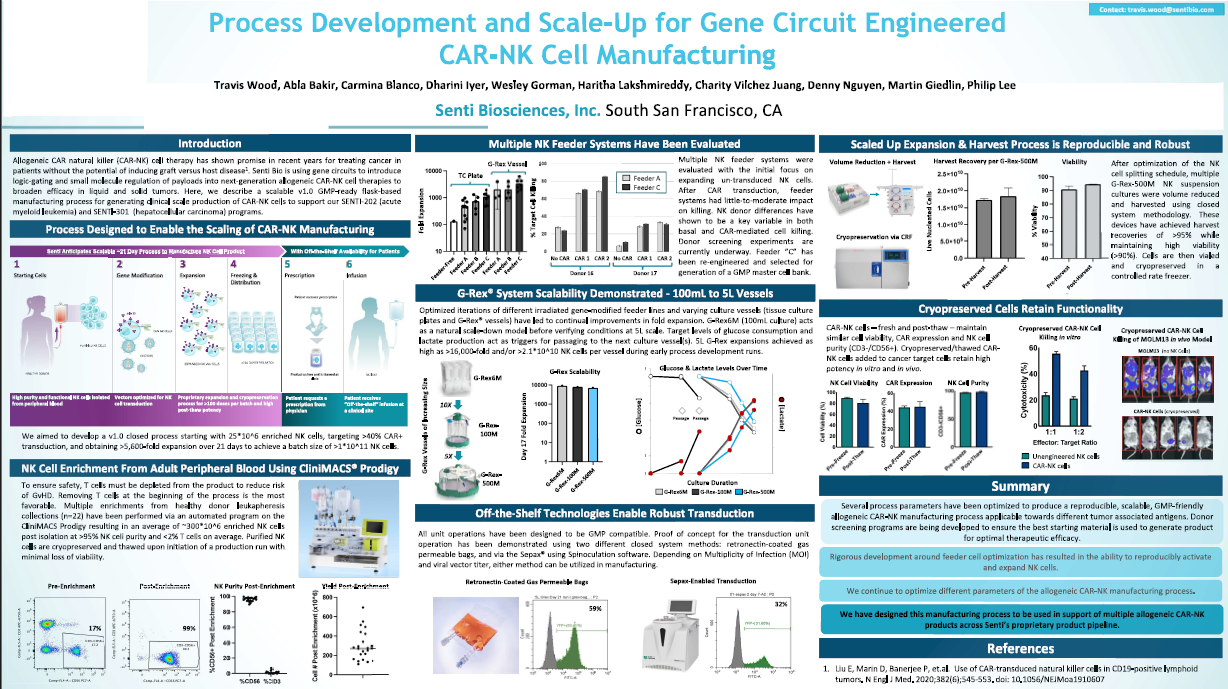
G-Rex® Grant Tour
Supporting the Emily Whitehead Foundation
MADRID
Location
Palacio Neptuno
Calle Cervantes 42, 28014, Madrid, Spain
Date
Friday, June 05, 2026
12:00PM - 7:00PM
Hear from leaders bringing hope to patients through advanced cell & gene therapies

Meet Dr. Alena Gros Vidal

Meet Dr. Begoña Diez Cabezas

Meet Dr. Núria Gavaldà

Meet Mr. Ignacio Nunez, M.Sc., MBB
Global Resources, Madrid Innovation.
Join the coalition accelerating patient cures right here in Madrid.
Tackling Solid Tumors
Dive deep into 'The Last Frontier' of cancer therapy with pioneers from Vall d'Hebron Instititute of Oncology
State of CGT in Madrid
Engage with experts shaping the future of the Spain biotech ecosystem.
Network and Engage
Participate in the "Believe Bundle Creation" and support the Emily Whitehead Foundation’s mission to save lives.
Event Sponsors








Event Essentials
Prepare for a full day of innovation, networking, and impact.
Mark your calendar
10:00 AM - 7:00 PM
G-Rex Grant Tour
If you're building the future of cell therapy - this is your place
Interested in Sponsoring?
Are you a provider of tools, technology, or services used in research, development, or manufacturing of cell and gene therapies?
Does your innovation pair nicely with G-Rex?
If yes, please consider sponsoring one, some, or all of the G-Rex Grant Tour events. Your sponsorship is a tax deductible donation to the Emily Whitehead Foundation.