Share
G-Rex devices have been used in the development and manufacturing of CAR-T cell therapies for over 10 years. Notable early adopters include Baylor College of Medicine’s Cell & Gene Therapy, NCI Surgery Branch, and the Seattle Children’s Research Institute. Moreover, G-Rex is used in commercial production of the most successful, profitable, and accessible CAR-T cell therapy on the market.
Contrary to popular belief, a G-Rex device can (and should) be used to perform the majority of the core unit operations in canonical CAR-T cell production: Purification, Activation, Transduction, Expansion, and Concentration.
The need for space-efficient, high-throughput manufacturing is a requirement of all modes of CAR-T cell therapy production, including distributed production. It matters not if a developer chooses to manufacture from one centralized facility or many de-centralized facilities, high-throughput manufacturing will be a common denominator. Because of this, many institutions leading the charge on decentralized manufacturing of CAR-T cell therapies have turned to G-Rex. One of the leading developers and manufacturer’s of localized CAR-T cell therapy production is Caring Cross.
Caring Cross is a 501(c)(3) non-profit focused upon accelerating the development and implementation of advanced medicines like CAR-T cell and stem cell gene therapies. In partnership with like-minded stakeholders, Caring Cross develops and deploys technologies, systems, and therapeutic candidates that improve the affordability and accessibility of these potentially life-saving therapies for all patients in need, everywhere.

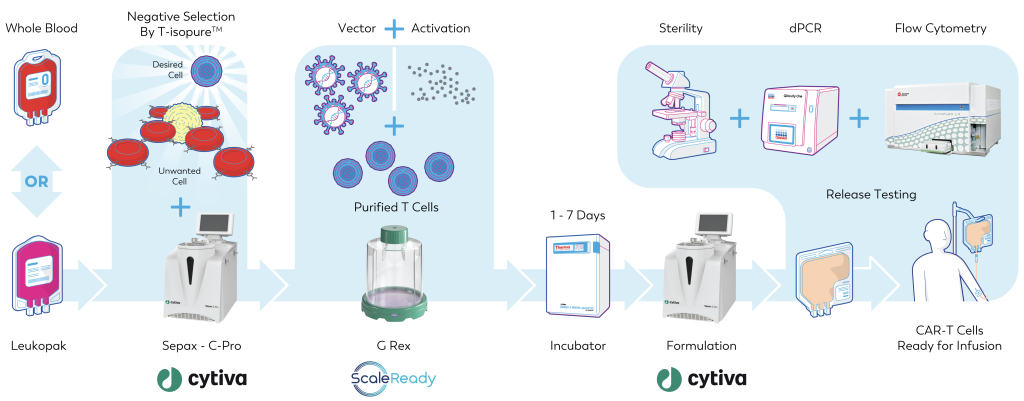
As shown, G-Rex is the bioreactor of choice for Caring Cross as a way “to build a low-cost CAR-T point-of-care production pipeline, at scale, that can be implemented in a dedicated clean-room or in a mobile clean-room setting”.
G-Rex bioreactors are available in fully closed formats with validated sterile fluid paths. Not only does this enable manufacturing outside of the biosafety cabinet in less controlled clean rooms, it also enables a multi-product ballroom concept.
Closed system G-Rex bioreactors have the smallest footprint in the industry and this enables the most economical use of space.
Fluid can be inoculated into standard G-Rex through simple sterile tube welding and heat sealing. Future directors of G-Rex product development efforts will seek to eliminate tube welding and sealing altogether in favor of sterile connectors.
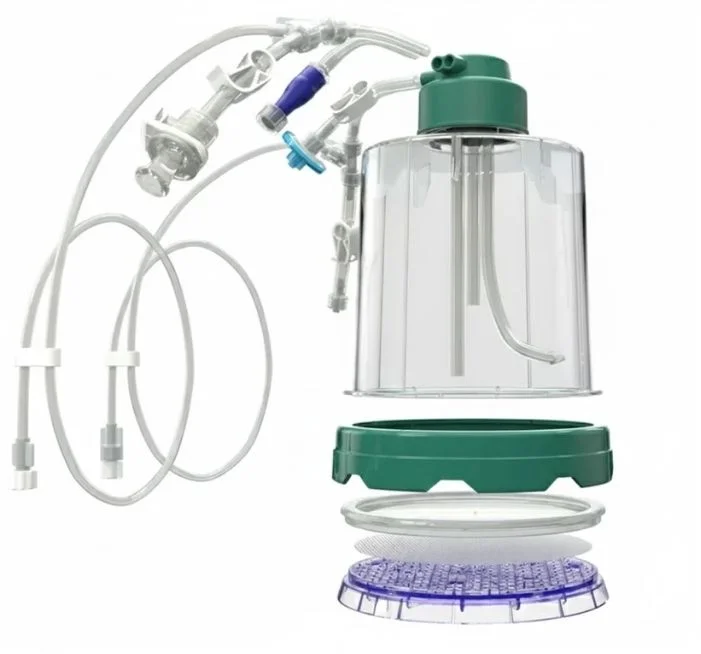
Moreover, it has recently been demonstrated that complete elimination of the biosafety cabinet can be achieved using clever new products from our collaborators at Biolife Solutions.
Eliminating the biosafety cabinet is a critical achievement that removes bottlenecks in manufacturing, dramatically streamlines the flow of the drug substance through the final assembly line, directly increasing throughput and lowering cost of goods. This marks a major step forward towards increasing global patient access to advanced therapies by lowering the burden to implementing highly efficient G-Rex based CGT production.
Let’s talk more about how ScaleReady can advance your cell and gene therapy research and manufacturing with ingenious efficiency.
