Miltenyi Biotech’s CliniMACS Prodigy is the gold standard “all-in-one” or “black-box”* bioreactor where a single-machine completes the full CAR-T cell therapy assembly process. Apheresis in, final drug product out.
Miltenyi Biotech’s CliniMACS Prodigy is the gold standard “all-in-one” or “black-box”* bioreactor where a single-machine completes the full CAR-T cell therapy assembly process. Apheresis in, final drug product out.
The CliniMACS Prodigy is functionally closed, mostly automated, and provides repeatable and reliable drug product manufacturing. It is used to make a variety of cell and gene therapy drug products (CAR-T, HSC, etc.) in a variety of manufacturing models (centralized, distributed, point-of-care, etc.). Standardized protocols are offered with the ability for “custom application” development and Miltenyi offers best-in-class reagents that are tailormade for use in their machine.
The CliniMACS Prodigy really is a marvel of modern engineering and few innovators have done more to advance the field of Cell & Gene Therapy than Miltenyi Biotech and their MACS technology.
"The most dangerous form of waste is that which we do not recognize"
- Shigeo Shingo
Nonetheless, there are two (2) core engineering limitations that the CliniMACS Prodigy cannot overcome without being fundamentally redesigned into a new machine:
- An inability to disconnect the drug substance from the Isolation and Formulation Units
- The requirement to force-feed media (nutrients) and oxygen to the cells
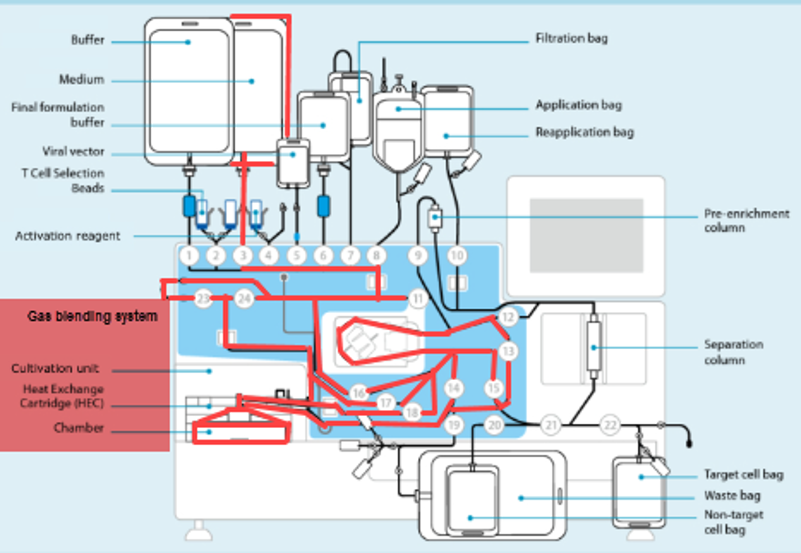
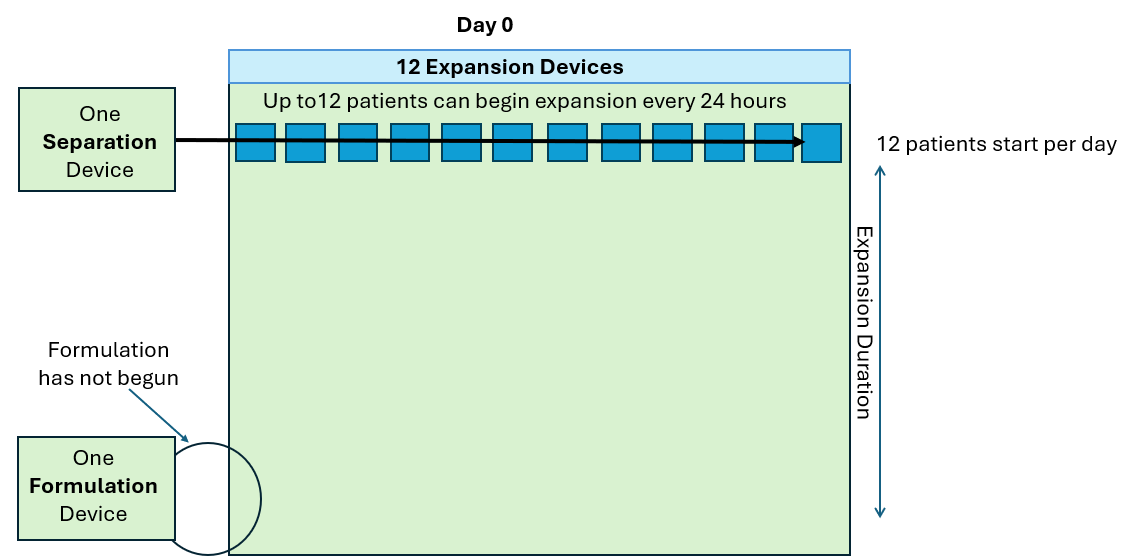
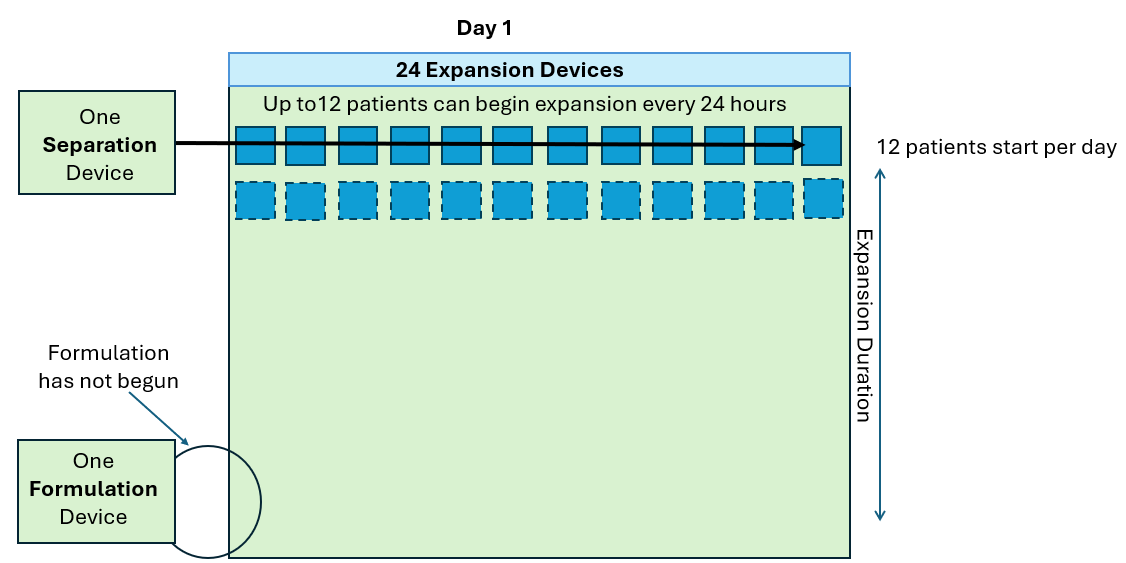
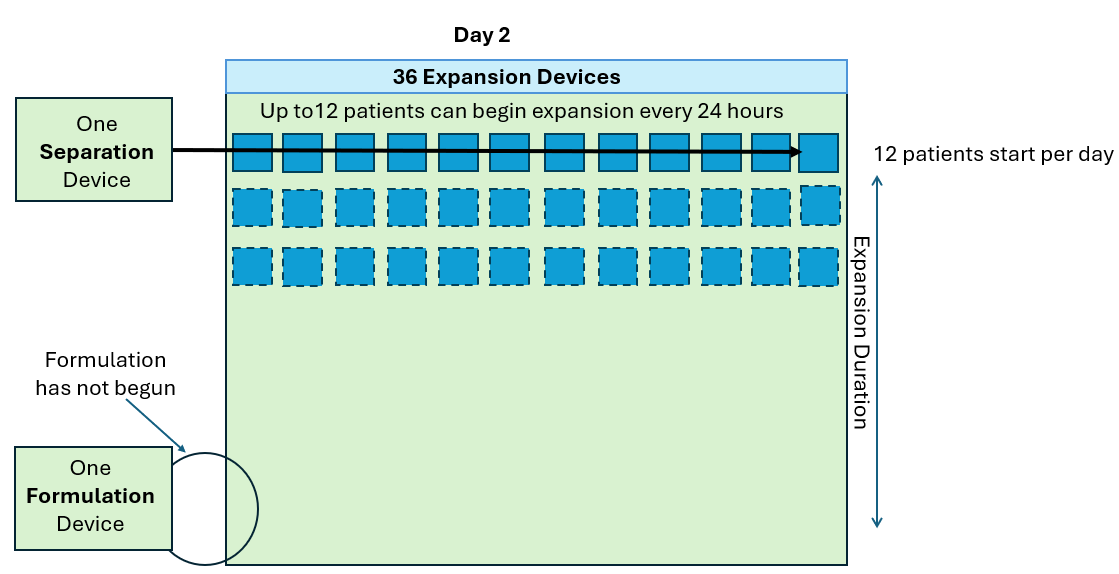
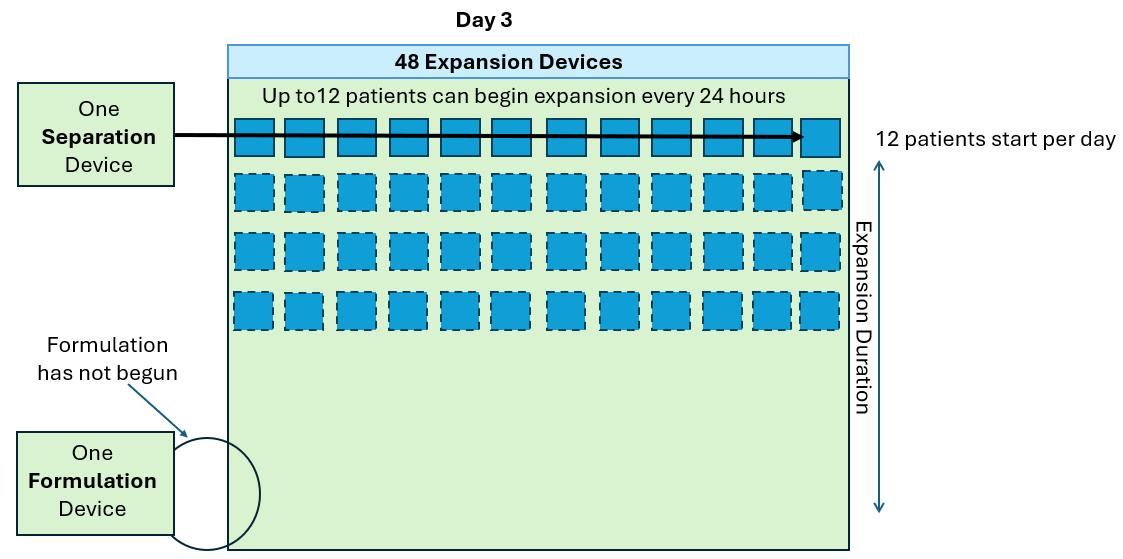
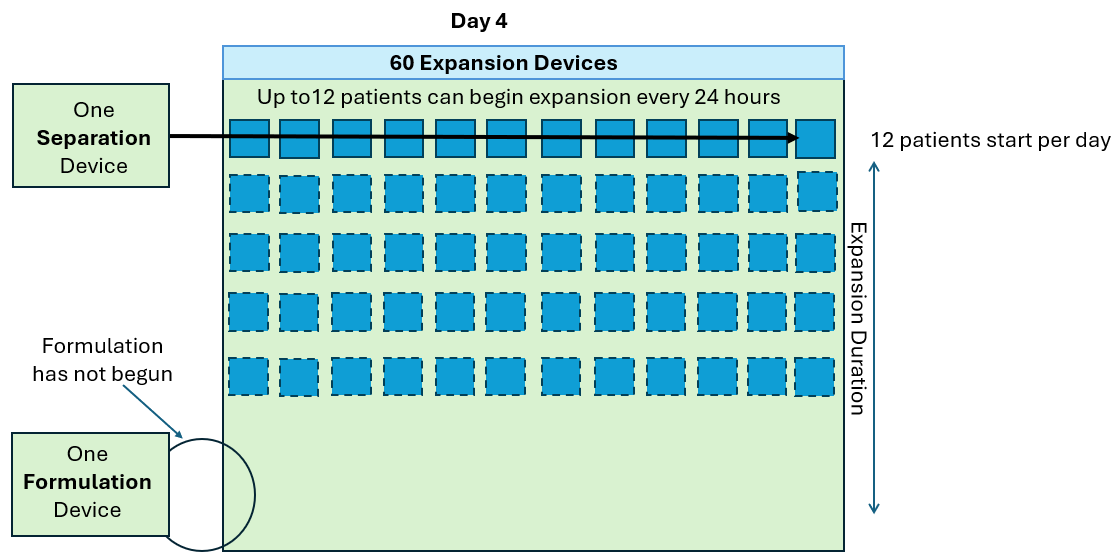
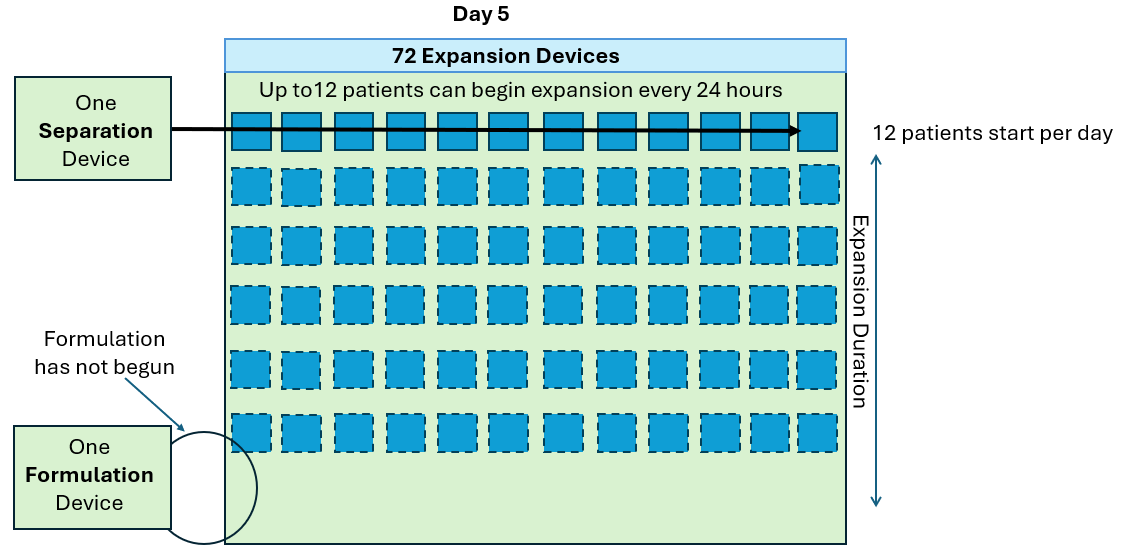
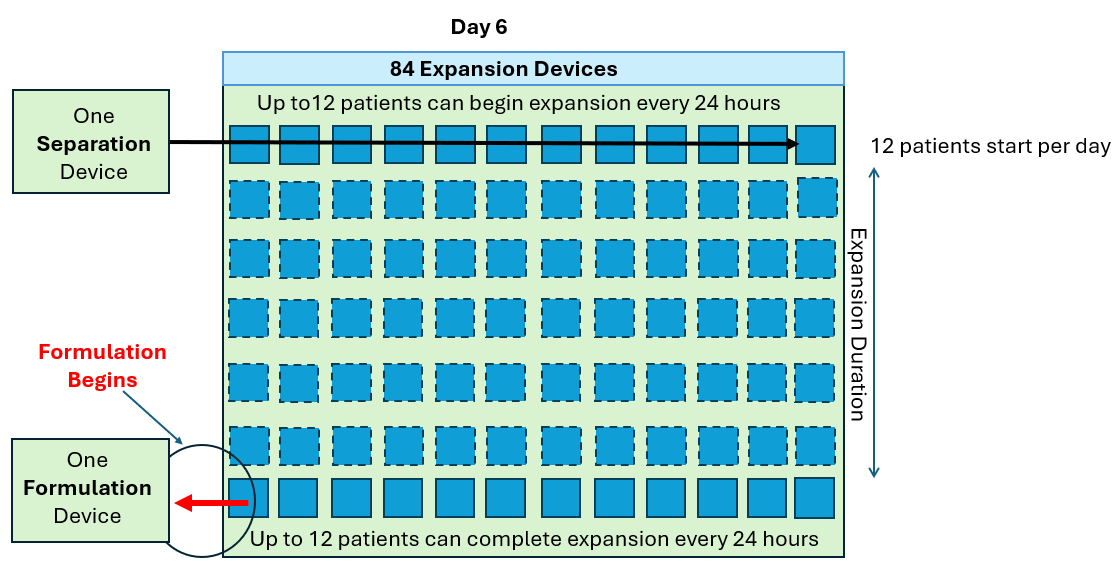
As seen in the annotated patent drawing, the selection instrument (a critical asset) remains idle once the drug substance undergoes T cell isolation. Similarly, the formulation unit (also a critical asset) remains idle as well while it awaiting final assembly of the drug product. This is because the Prodigy’s dedicated single use consumable maintains a continuous fluidic connection to the drug substance, relegating the machine to in-series batch production (i.e. only one drug product at a time).
In this scenario, critical assets used in the Selection and Formulation unit operations remain idle for anywhere between 96-99% of the full assembly process. The waste of waiting (or idleness) has long been identified as one of the core wastes in LEAN manufacturing and the “all-in-one” assembly process is a prime example of this.

"There is nothing so useless as doing efficiently that which should not be done at all"
- Peter Drucker
Thus, it matters not how efficiently a single machine can perform the all-in-one drug product assembly when it should not be done at all.
Why should “all-in-one” assembly not be done at all? Because the waste associated with the idle equipment restricts this approach to extremely low-throughput and high capital equipment and space costs. The bottleneck is the full manufacturing process itself.
In order to produce 12 drug products per day of the week (4,368 patients per year), let’s look at how many CliniMACS Prodigies would be required in a standard 7-day manufacturing process.
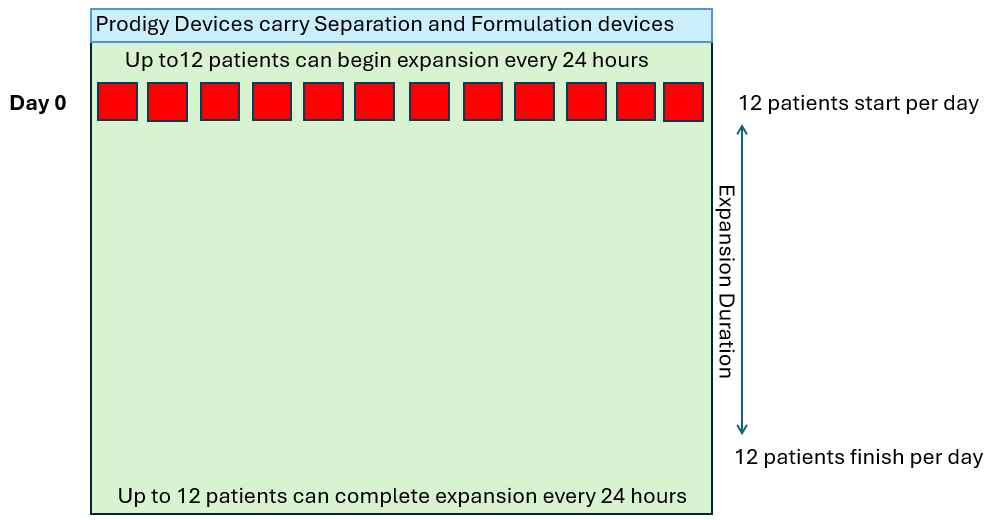
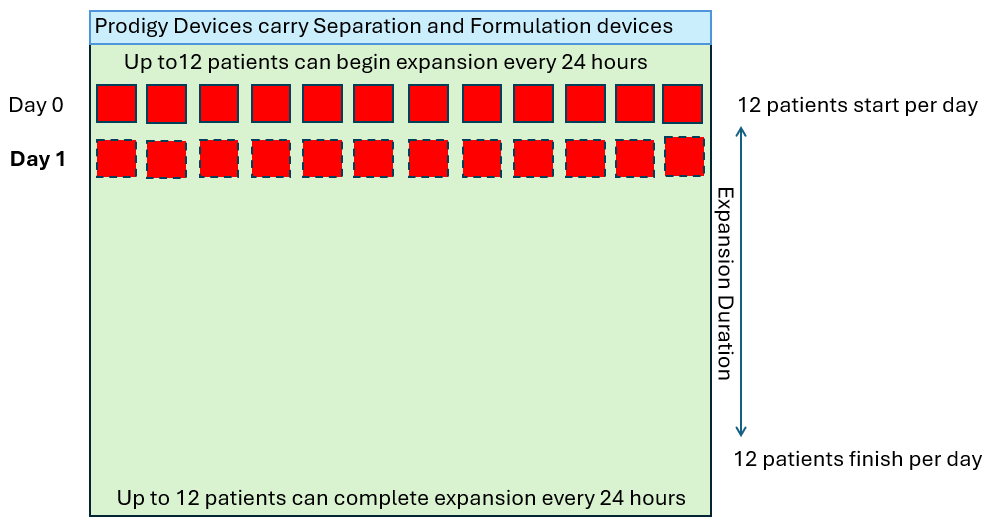
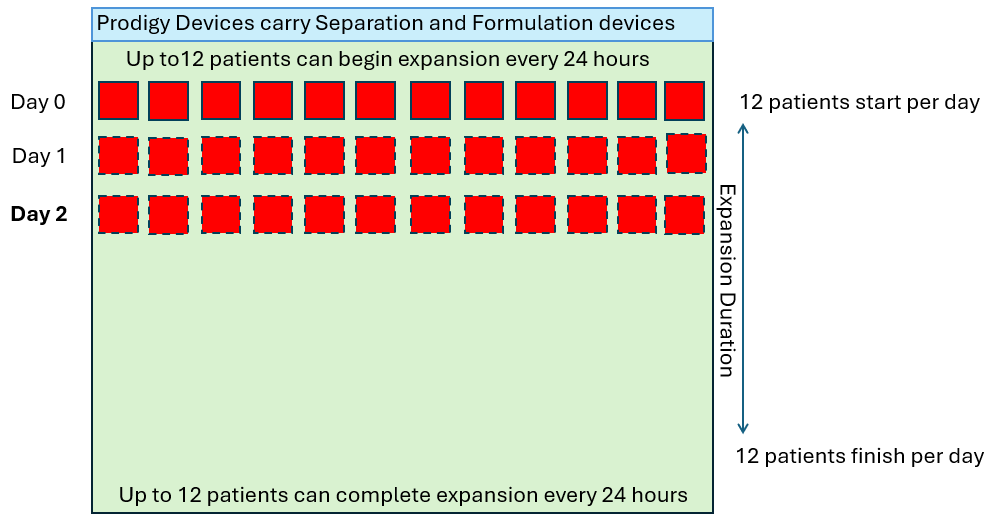
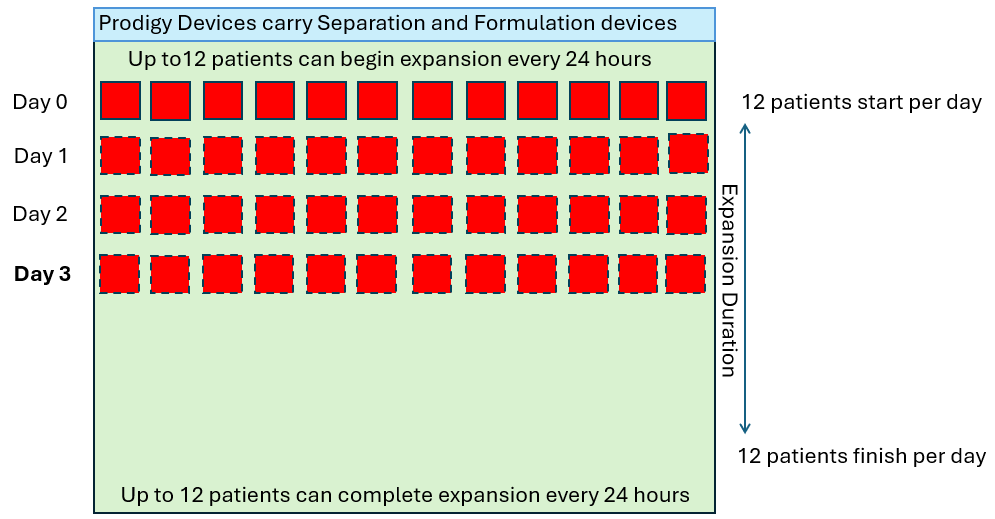
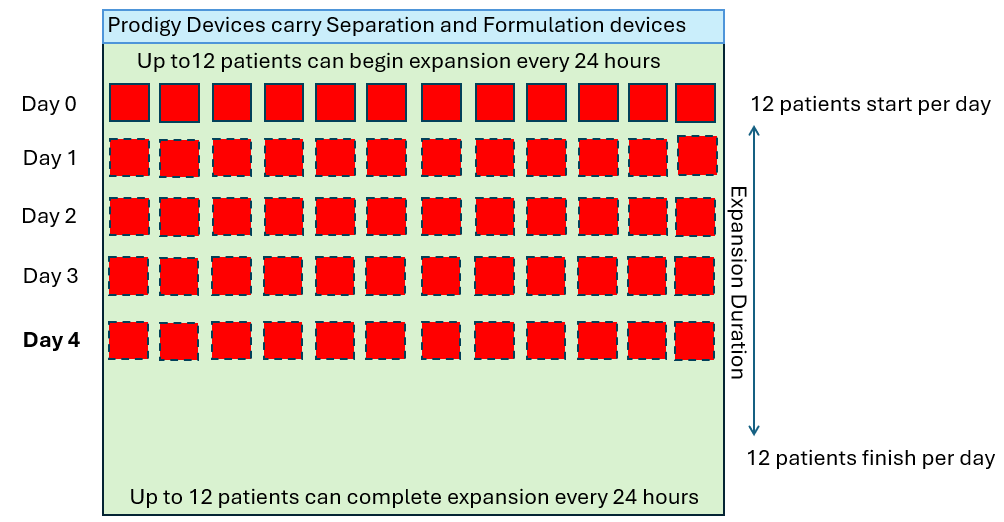

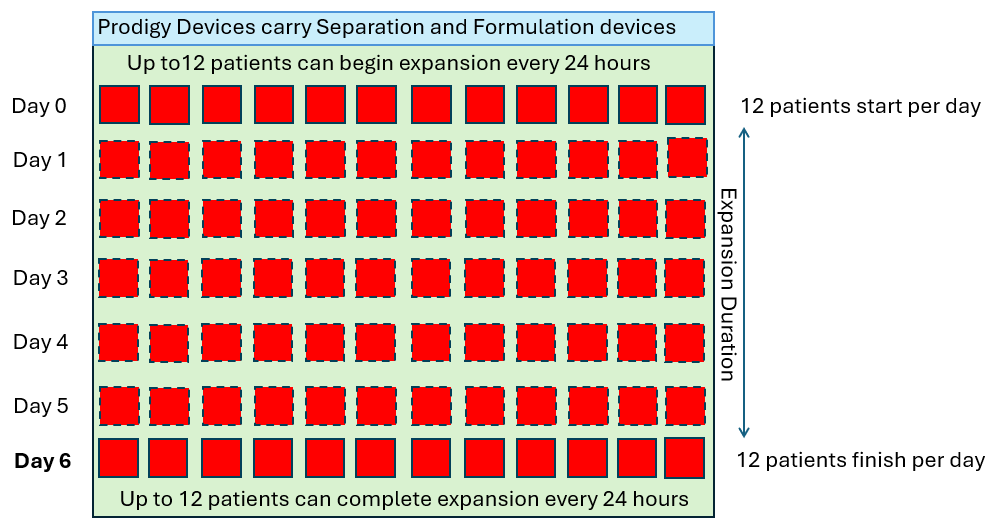
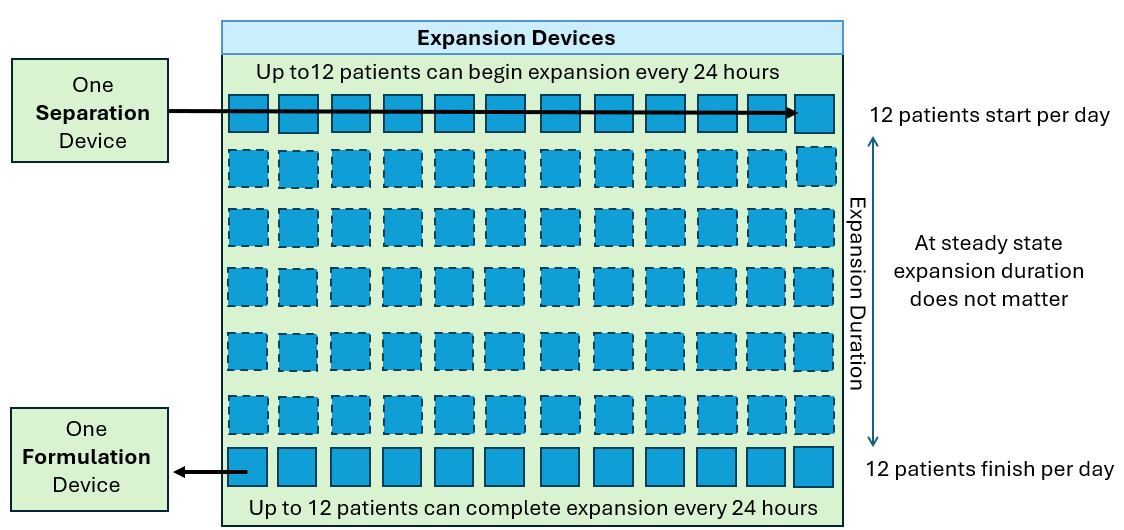
As we see above, the quantity of CliniMACS Prodigy instruments required is proportional to the throughput requirement and the duration of the process. However, in these examples, it is assumed that the demand is linear (twelve per day every day) but in reality this is not how patients are referred. The demand will fluctuate up and down in some sort of wave pattern as shown below.
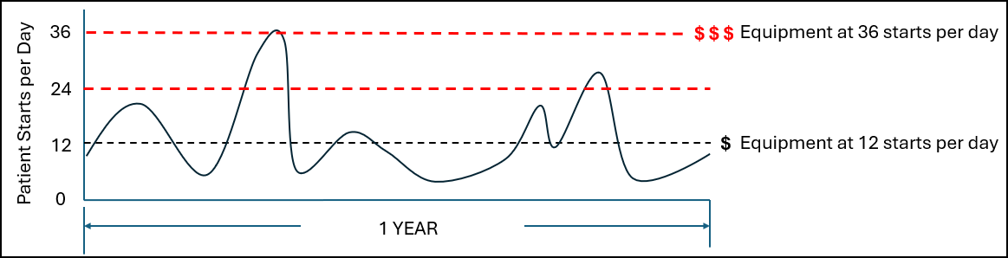
Much like a restaurant builds capacity for the Friday night surge demand, so to must developers of autologous cell therapies build capacity for surge demand of patients. Thus, 2-3x additional CliniMACS Prodigy instruments would be required to ensure sufficient spare capacity so advanced therapies are delivered just-in-time to all patients.
(# of starts per day) x (process duration) x (spare capacity coefficient) x (est. cost of CliniMACS Prodigy) = Total Cost
12 x 7 x 3 x $375,000 = $31,550,000 (plus recurring ~10% annual service fee per year)
How does this compare to a modular G-Rex approach?
In a modular G-Rex approach, the drug substance can be connected and disconnected from bespoke instruments designed for each step and the bottleneck becomes the longest cycle time of the individual unit operations.
For example, assume it takes an operator two (2) hours to set up the isolation machine (e.g. install tubing set, load reagents, program software, etc.), run the isolation, and perform line clearance. In this scenario, every two (2) hours a new culture vessel is inoculated for activation, transduction, and expansion.