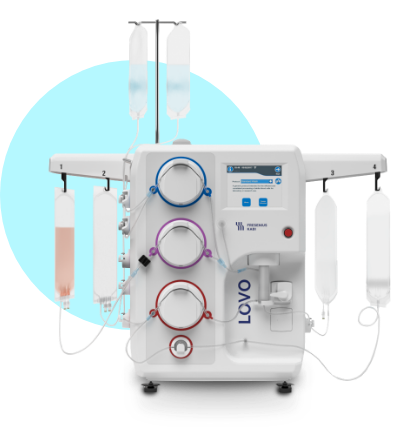
Lovo®
The First Step Toward High-Throughput Cell Processing.

Achieve flexibility and scalability in a single instrument.
Lovo’s flexibility supports multiple applications, at any stage of your operation to support each unique cell therapy manufacturing process. Whether you’re working with millions or billions of cells, in 10mL or 22L, Lovo adapts with you to easily scale your process, from Phase 1 through Commercialization.
Process fast. Maximize control.
Only Lovo delivers automated, functionally closed cell processing through fast, adaptable, non-fouling spinning membrane filtration.
You don’t need to trade cell recovery with washout efficiency. Lovo’s spinning membrane filtration allows you to process a wide range of cell volumes and concentrations quickly while maximizing recovery and cell viability.
Lovo Instrument Training Program
Training as flexible and as agile as your CGT manufacturing process.
- Flexible training options
- Self-paced e-learning courses offered through our Global Learning Academy
- Hands-on Training is offered at our U.S. Learning Center and led by expert industry instructors
- Custom training courses can be tailored to specific learning and development needs
- Gain expertise in Cell Processing instrument operations
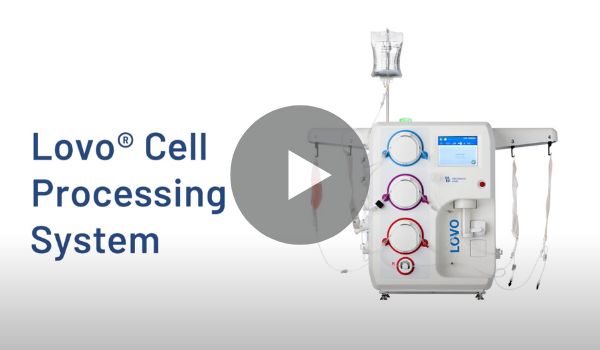
Discover Lovo.
Lovo blends flexible, customizable features to meet your current needs and adapt to future requirements.
Lovo’s intuitive operation simplifies your processes while minimizing operator interaction. Easily configure parameters and create protocols for different cells, different applications, and different trials — all on the same platform. Then lock down for manufacturing, and scale up as your operation evolves


Access Secure Data Anytime. Anywhere.
DXT allows for secure, wired or wireless data transfer. Generate reports, filter procedure records, and export directly to Microsoft Excel or your LIMS.
Securely transfer data directly from your Lovo via wired or wireless network. Generate non-editable, downloadable, or printable procedure records. Create filterable procedure records for easy searching. Export data directly to Microsoft Excel or LIMS.
Ready to Discover Lovo?
Testimonials
Associate Director, Analytical Development
Large Life Sciences Group
Additional automated cell processing resources
Lovo Cell Processing System Brochure
- Instrument features and design
- Applications supported
- Customizable protocol design
DXT Data Management System for Lovo
Cue Cell Processing System Brochure
- Instrument Features
- Applications supported
- Customizable protocol design
See how Lovo fits into the ScaleReady™ workflow.
The Lovo and Cue Cell Processing Systems are for laboratory use only and may not be used for direct transfusion. Appropriate regulatory clearance is required by the user for clinical use.
For applications requiring regulatory clearance or approval, Users may request the required technical documentation from Fresenius Kabi to support their submissions.
Refer to the Lovo Cell Processing System Operator’s Manual, Cue Cell Processing System User’s Guide, DXT Administrator’s Guide and DXT User’s Guide for a complete list of warnings and precautions associated with the use of these products.